Intake Ports
Right Cylinder
BEAU de ROCHA FUNCTIONS / ATTRIBUTES
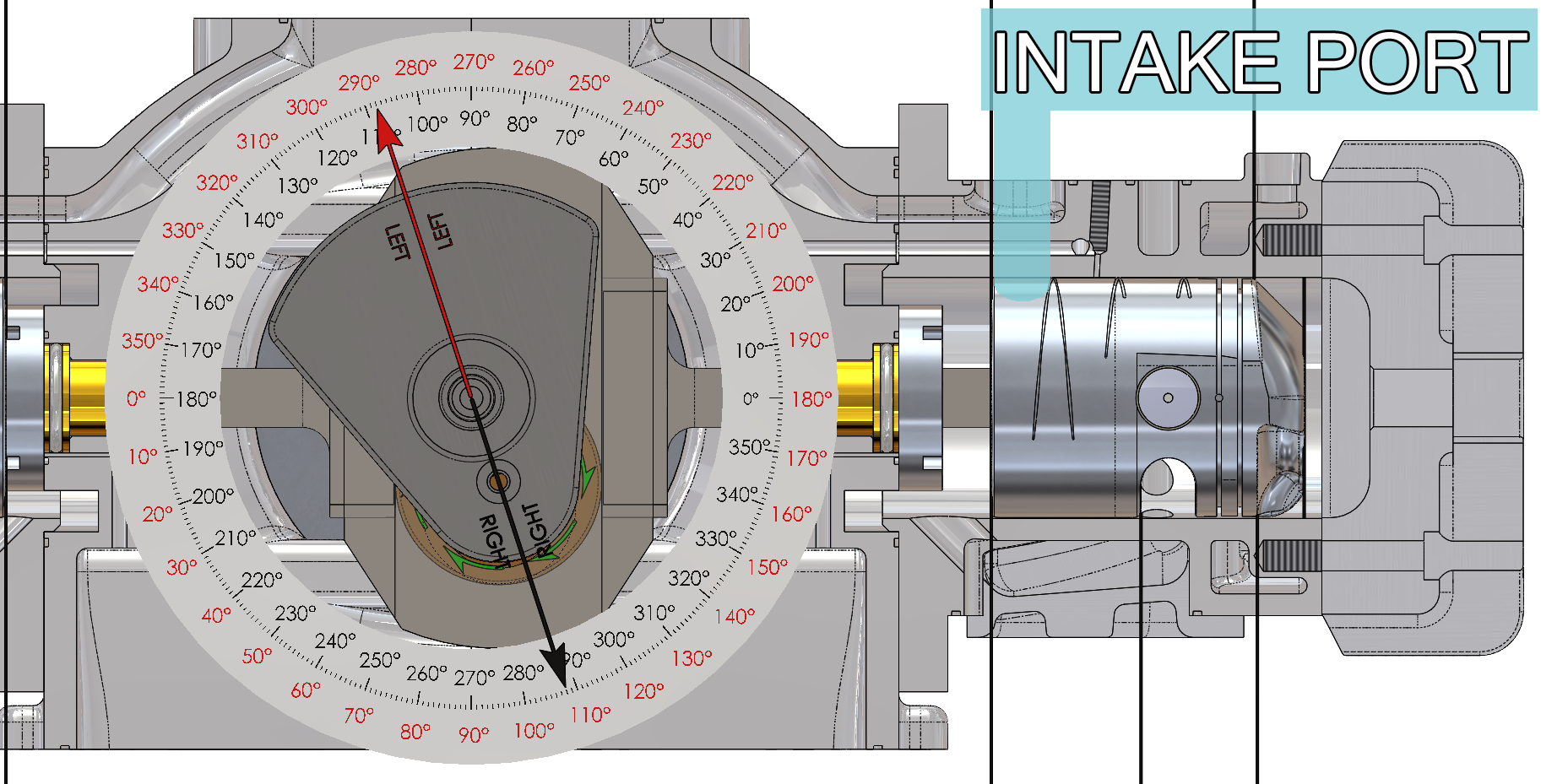
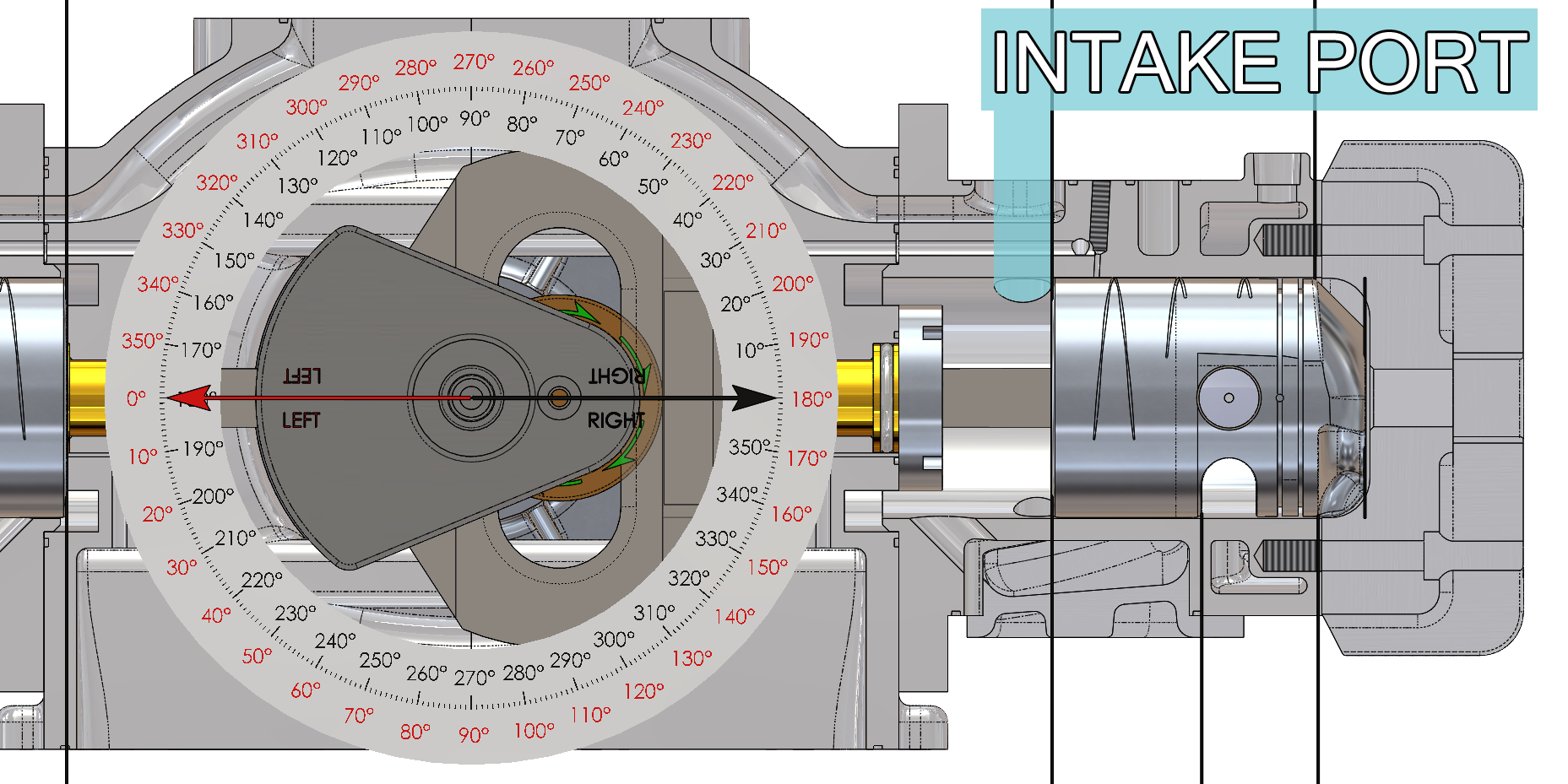
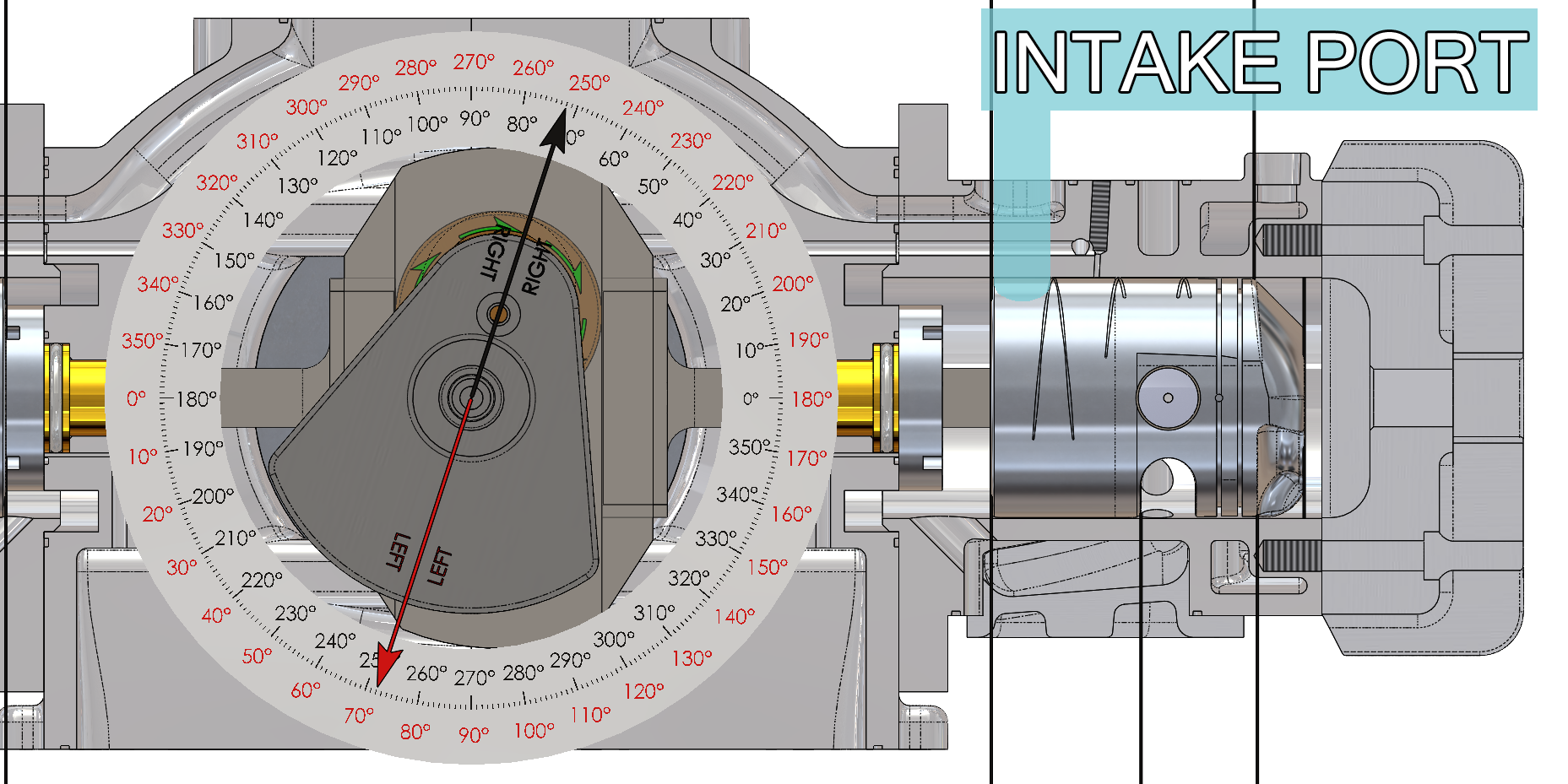
The intake ports are opened by the piston skirt for 144 degrees of crankshaft travel (from 288 degrees before top dead center until 72 degrees after top dead center – black arrow and black degree compass on the animation) and the area underneath the piston and the crankcase facing is filled with fresh air/fuel mixture. The following function that Beau de Rocha identified in 1862 has been accomplished:
- Intake (under the piston).
RUSSELL BOURKE’S FORMULA
- Port openings will always be computed from the degree of crankshaft travel.
- Intake ports open 72 degrees before top dead center (288 degrees – black arrow and black degree compass on the animation) and close 72 degrees after top dead center (72 degrees – black arrow and black degree compass on the animation).
- The intake ports are open for 144 degrees of crankshaft travel (from 288 degrees before top dead center until 72 degrees after top dead center – black arrow and black degree compass on the animation).
- Intake ports equal exhaust ports in area. See the Bourke formula (Table II) for the exhaust port hole size calculation (four holes) and match the intake port hole size area (two holes for the animation) to the exhaust port hole size area.
- That portion of the piston in contact with the cylinder wall on the exhaust and intake port side must be the same diameter as the cylinder bore when the engine is hot at running temperature. This rule is extremely important to ensure there is no leakage from the intake and exhaust ports. It is also critically important for oil control as this engine will run on motor oil, excess oil will upset the air/fuel ratio and/or you will not be able to turn the engine off.
OTHER IMPORTANT INFORMATION
The fuel injection system needs to atomize the fuel as much as possible. The air/fuel mixture requires complete fuel vaporization and complete homogenization. Three (3) things that the Bourke Engine does to insure complete vaporization and complete homogenization are:
- Expose the fuel and the air/fuel mixture to heat
- Heated fuel and heated air/fuel mixture are exposed to a partial vacuum. A heated liquid fuel will flash to vapor if the fuel is hot enough and/or the vacuum is strong enough.
- The air/fuel mixture is exposed to violent turbulence.
As demonstrated in the detonation demo, the quality of the charge is far more important than the quantity of the charge. The quantity of the charge in a conventional internal combustion engine (ICE) is the determining factor for how much power that engine will produce. In the Bourke Engine, the detonation of even a small quantity of lean, homogenized air/fuel mixture produces tremendous power (based on the detonation demonstration, up to 500 times more powerful than progressive burning? – we challenge the scientific community to tell the world how much more powerful detonation is!!!)
All of the volumes shown below for the 10.842 cubic inch (177.6748 cc) model were calculated using Solid Works:
- The volume under the piston when it is at Top Dead Center (TDC) is 166.478 cubic centimeters (cc), or 10.1591 cubic inches.
- The volume under the piston when it is at Bottom Dead Center (BDC) is 88.7875 cc, or 5.4181 cubic inches.
- The volume in the combustion area when the piston is at Bottom Dead Center (BDC) is 88.8374 cc, or 5.4212 cubic inches.
- The volume in the combustion area when the piston is at Top Dead Center (TDC) is 7.2358 cc, or 0.4416 cubic inches.
The compression ratio for this model is calculated by dividing the volume at BDC by the volume at TDC (88.8374 cc/7.2358 cc =12.28:1 compression ratio).
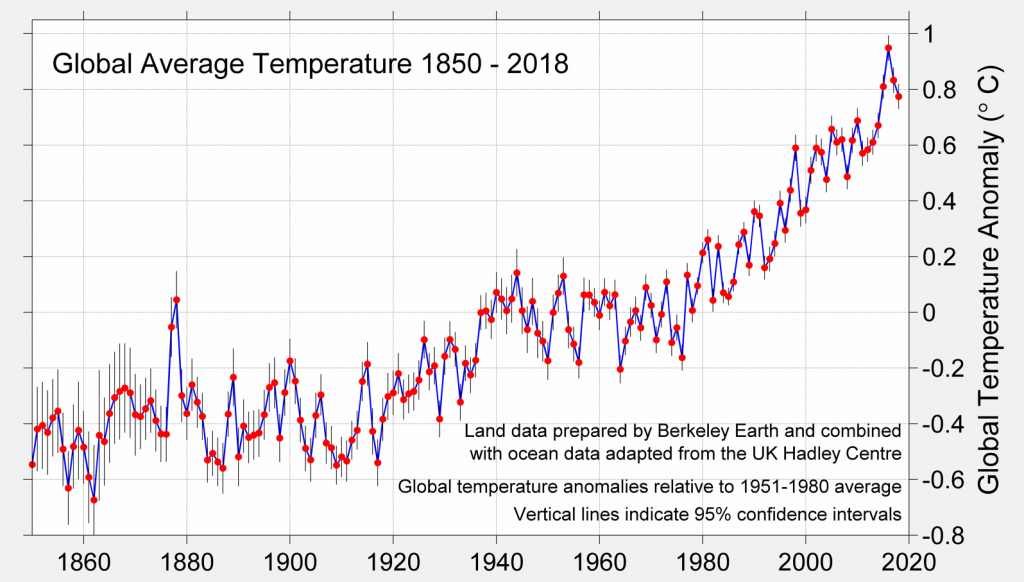
Global Warming
The chart shows the increase in global average temperature from 1850 to 2018.
Source: Berkeley Earth
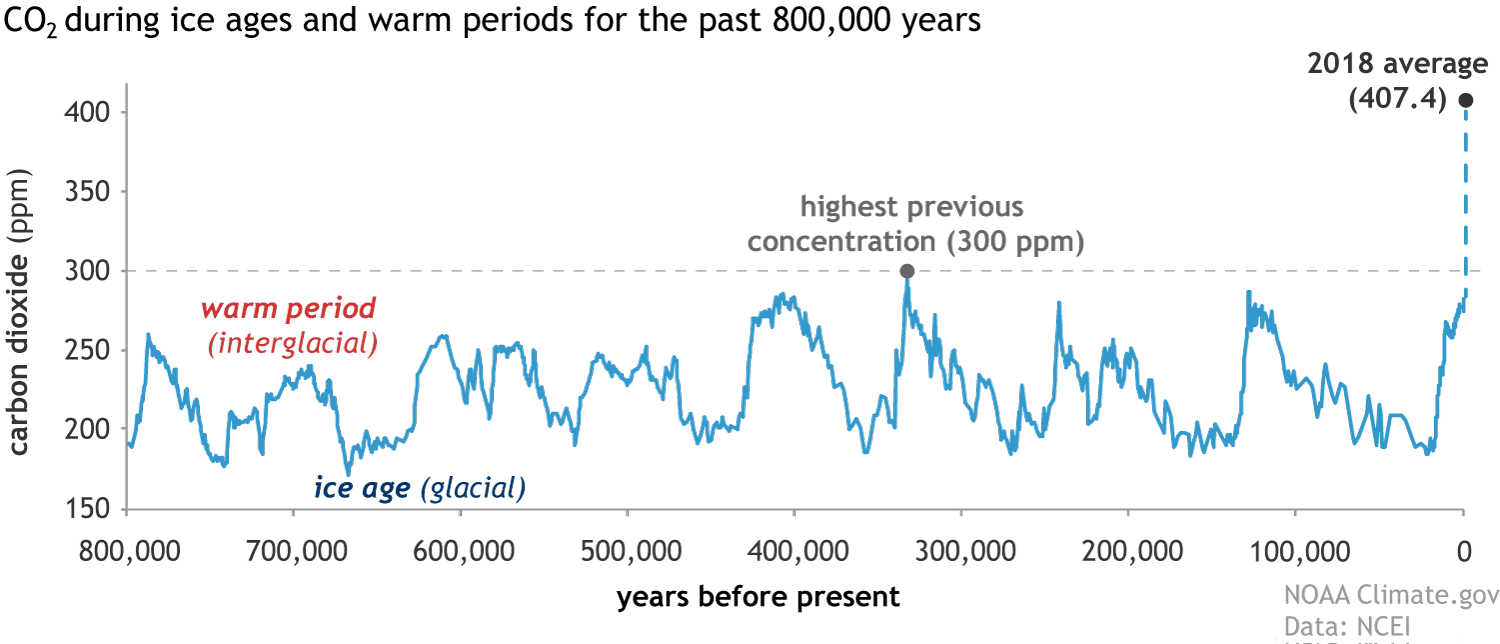
Carbon Dioxide In Our Atmosphere
Read all of the scientific reports!!!!!! We are at 415 parts per million, more than anytime in the last 800,000 years. The Bourke Engine using fuel from algae for all transportation and distributive power generation will reduce our carbon footprint for these activities by AT LEAST 85%.

WAKE UP WORLD, WE ARE KILLING OUR PLANET!!!!
2019 – Was the Second Hottest Year on Record!!!!
Global Ocean Acidification
As our oceans absorb excess carbon dioxide, the carbon dioxide reacts with the sea water forming carbonic acid which is changing the pH of our oceans making them more acidic. Research is ongoing as to how the increased acidic level of our oceans is affecting corals and marine life.
Source: IPCC 2007
©2019 Copyright Bourke Engine Project, LLC
All Rights Reserved. All of the information contained within this website can be used, with permission, by any other website providing that the requesting party agrees to the terms and conditions as described in the PERMISSION FORM.
© Copyright 2019-2023 Designed by Lynn